Analyzing the composition and grade of foundry metal
Spectrochemical analysis is a type of chemical analysis used to determine the arrangement of atoms and electrons within molecules of chemical compounds. It observes the amount of energy absorbed during changes in motion or structure. The wavelength and intensity of electromagnet radiation is measured to produce quantifiable results that are primarily used for quality assessment.
Spectroscopy and the spectrometer
Spectroscopy and spectrometer are terms that appear often when discussing spectrochemical analysis. Put simply, spectroscopy is the study of energy in relation to a sample material, and a spectrometer is the instrument used during spectrometry, the act of spectroscopy.
Spectroscopy
Spectroscopy is the study of the interaction between radiated energy and a sample material. This interaction produces electromagnetic waves in the form of visible light, typically seen as sparks. Spectroscopy was introduced in the 17th century when Sir Isaac Newton discovered that white light could be separated into component colors using a prism, and these components could be recombined to form white light. He realized that the prism is not what creates the colors, but it instead works to separate component colors of white light. In the early 1800s, Joseph von Fraunhofer performed experiments that further evolved spectroscopy into a more precise and quantitative scientific technique. However, it was not until the 19th century that the quantitative measurement of dispersed light was standardized and recognized as a sound method of testing.
Spectrometer
A spectrometer is the instrument used in spectroscopy that produces spectral lines and measures their wavelengths and intensities. It is a scientific device that separates particles, atoms, and molecules by their mass, momentum, or energy. Spectrometers are integral to chemical analysis and particle physics. There are two types of spectrometers: optical and mass spectrometers.
Optical spectrometer
An optical spectrometer, or simply “spectrometer,” is able to separate white light and measure individual narrow bands of color (spectrum). It shows the intensity of light as a function of wavelength or frequency and the deflection is created by refraction in a prism, or by diffraction in a diffraction grating. Optical spectrometers use the concept of optical dispersion—and since each element in a sample leaves a unique spectral signature, spectral analysis can determine the composition of the sample itself. Optical spectrometers are common in astronomy, metal production, solar power, and semiconductor industries.

Mass spectrometer
A mass spectrometer measures the spectrum of the masses of atoms or molecules that exist in a solid, liquid, or gas. It achieves this by measuring the mass-to-charge ratio and abundance of gas-phase ions. Mass spectrometers are used in the fields of pharmaceutical science, biotechnology, and geology.

Why is spectrometry needed?
Spectroscopic techniques are at the forefront of many technical fields. Spectrometry is needed for its role in research and development, as well as for its more practical role in material analysis for various industries. Science and technology have always relied on spectrometry—from early studies to advancing technologies that fuel modern research.
Radio-frequency spectroscopy led to magnetic resonance imaging (MRI), a ground-breaking, medical instrument used to visualize internal soft tissue of the body. Radio and x-ray spectroscopy paved the way for astronomical research on distant stars and intergalactic molecules. Optical spectroscopy is routinely used in industrial and environmental settings to identify chemical composition of matter. Without this application in spectrometry, today’s fast and effective method of alloy identification and material inspection would not exist.
Optical emission spectroscopy
Optical emission spectroscopy (OES) is a common form of spectroscopy used to determine elemental components in solid metal samples. It is widely used in foundries and metal production facilities because it can analyze a vast range of elements with high precision and accuracy. The sample metals used in OES can be from the melt in primary and secondary metal production, or processed metals such as rods, plates, wires, and bolts.

How does optical emission spectroscopy work?
OES provides quantitative analysis using three key components: an electrical source, an optical system, and a computer system.
1) Electrical source
An electrical source is required to excite atoms into an active state within a metal sample. A small portion of the sample is heated to thousands of degrees Celsius using a high voltage electrical source in the spectrometer through an electrode. An electrical discharge is produced due to the difference in electrical potential between the electrode and the sample metal. This electrical discharge causes the sample metal to heat up and vaporize at the surface.
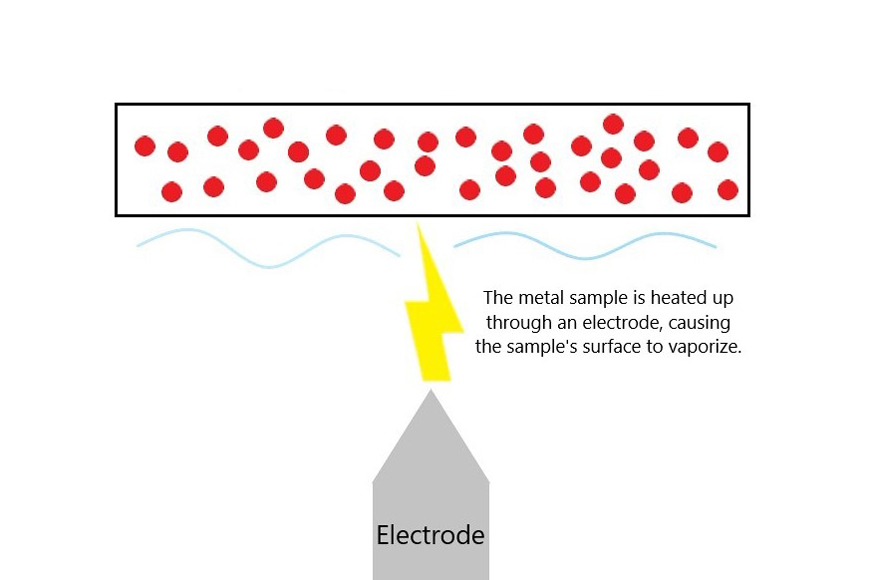
During this process, the activated atoms produce emission lines that are distinct to each element. Two types of electrical discharges exist: an electric arc or spark. An electric arc produces an ongoing electrical discharge, much like lightning. An electric spark is more of an abrupt electric discharge—a brief emission of light often accompanied by a sharp snapping sound.
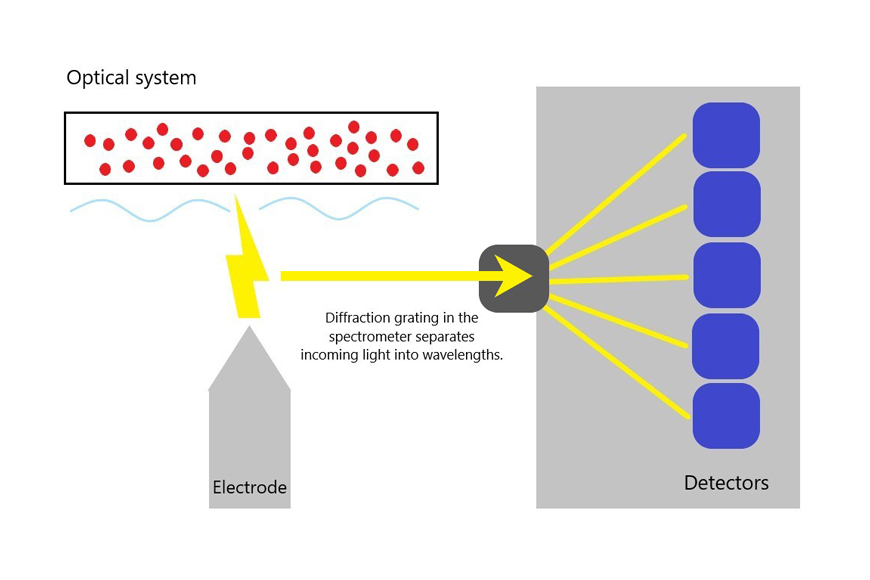
2) Optical system
The optical system transfers the emission lines from the vaporized sample, known as plasma, into the spectrometer. The diffraction grating in the spectrometer works to separate the incoming light into element-specific wavelengths. The intensity of light of each wavelength is then measured by a corresponding detector. The intensity measured during this process is proportional to the concentration of the element in the sample metal being tested. Since each element emits a specific set of wavelengths based on its electronic structure, the elemental composition can be determined by observing these wavelengths.
3) Computer system
Finally, a computer system is required to process the data. The measured intensities are processed through a predefined calibration to produce elemental concentrations. Modern technology has advanced the user interface to offer clear results with minimal operator intervention.
OES is user-friendly and widely accepted in the metal manufacturing industry. Although a popular instrument, it still comes with a few limitations including minor surface damage to the sample material and the need for constant maintenance.
OPTICAL EMISSION SPECTROSCOPY
Advantages
Disadvantages
- Fast quantitative determination of elements (typically less than one minute).
- Low capital investment and operating costs.
- Easy sample preparation.
- Quick analysis of carbon, nitrogen, oxygen, phosphorus, and sulfur in steel.
- Calculates carbon content (%) of stainless steels or low alloy steels.
- Distinguishes between 304/316 and 304L/316L stainless steels.
- Provides input data to carbon equivalency calculation.
- Not completely “non-destructive” (slight surface damage can be expected).
- Cannot test small parts (less than the size of a dime).
- Difficult to test in tightly confined spaces.
- Requires constant calibration and maintenance.
- Routine third party certification of results may be required.
Spectrometers in foundries
Optical emission spectroscopy can be employed on a range of materials from pure to alloyed metals. Foundries, as well as the aviation, automotive, and home appliance industries, benefit from spectrometers for process and quality control.
Spectrometers are often the instrument of choice for foundry metal analysis as they require only minimal intervention from foundry operators when used for inspection, quality control, and alloy identification. Stationary and portable versions exist, both with a high level of accuracy. Routine calibration and maintenance is required, and third party certification of results are often needed for spectrometer results to retain their validity. Spectrometers allow for metal analysis throughout the metal life cycle from metal production to processing, as well as at the end of its service life at recycling plants.













