Chemical layers that protect metal from corrosion

Many metals are vulnerable to corrosion when exposed to air and water. Corrosion can cause metal stress and part failure, so metallurgists seek ways to defeat it. One such method is metal passivation, a lesser-known method of corrosion-proofing a surface that uses a thin chemical layer as a seal. Passivation can occur naturally or be encouraged by manufacturing processes.
What is corrosion?
Metal corrosion occurs when molecules of an active metal alloy react in their environment to become electrochemically more stable. Oxides, hydroxides, and sulfides are the main compounds of corrosion. Simple exposure can lead to the reaction, like iron rusting in water and air. Electrochemical processes may also cause the reaction, as with the galvanic corrosion between nickel and cadmium in a battery.
There are metals that resist corrosion. Noble metals like gold, silver, and platinum are chemically stable in many conditions. Noble metals aren’t impervious to corrosion, but the process happens slowly, or with less common molecules. For example, silver’s thick black tarnish is usually caused by hydrogen sulfide rather than oxygen and water. Metallurgists check the active potential of metals using the galvanic scale or anodic index. Those near the top are less inherently prone to corrosion.
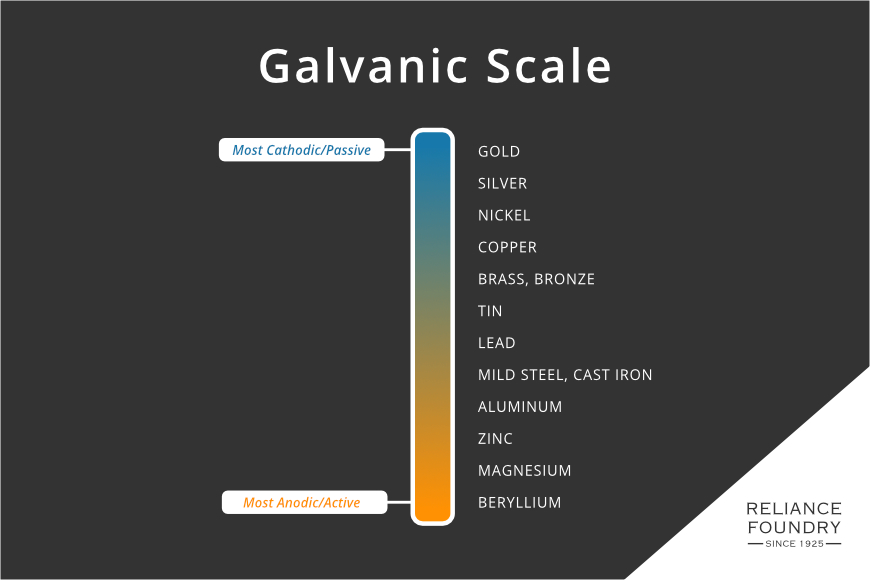
“Active” metals, or those nearer the bottom of the scale, are chemically less stable, and therefore more prone to reacting with other elements in the environment. To prevent this, active or passive processes are used to reduce corrosion. These processes generally work by “sealing” the metal with a top layer, preventing air and water from reaching the metal underneath. The layer can be manmade: paint, powder coating, and oil are all common sealants. If they are scratched, however, corrosion sneaks in.
Another option used to seal the metal is chemical, where chemical reactions are used instead of fabricated materials. One chemical process is passivation.
What is passivation?
Passivation, generally associated with stainless steel, is a treatment method to protect the metal from corroding through—making the material “passive” to the surrounding environment. Passivation, perhaps counterintuitively, encourages corrosion to occur on the surface, creating a thin layer of a new, non-reactive chemical. This top layer stays tightly bound to the metal, creating a natural seal that blocks the elements from corroding subsequent layers of the metal. A metal is passivated when every surface is covered with a tightly bound layer of corrosion. This layer may build naturally (passively) over time, but manufacturers can also actively induce it.

The history of passivation
Passivation was discovered by scientists doing electrochemical experiments, but they did not immediately recognize the utility of the process for general use. In 1790, chemist James Keir observed that a strong nitric acid bath did not corrode iron. He noted that when the same solution was water diluted, the iron corroded immediately, creating a bubbling solution of dark brown water. By 1836, Swiss chemist Christian Friedrich Schönbein specified the experiment further. He demonstrated that a piece of iron immersed in weak nitric acid dissolves and produces hydrogen, as Keir noted. Yet if the iron was first dipped into strong acid, it could then withstand the dilute acid. The corrosive elements of water seemed to be held at bay, at least for a while.
Michael Faraday, the British electrochemist, was the first to describe why. He hypothesized to Schönbein that an oxide skin created by the strong acid might cause the passive condition. As chemists and metallurgists explored the idea of a chemical “skin,” they sought techniques to manufacture or enhance passivation, and alloys that would create passive seals organically.
Passive oxide layers
As metals are exposed to the surrounding environment, if left unblocked by paint or powder coating, they will corrode naturally, creating the skin, or seal layer. Most passive films are made of oxides, combinations of metal and oxygen, so are called passive oxide layers.
One of the biggest benefits to passive oxide layers is that when they arise naturally, they will also naturally “heal” if the surface of the metal is scratched or otherwise damaged, as the next layer of molecules will then bond with the environmental elements.
The effectiveness of passive oxide layers depends on the type of elements involved. Not all oxide layers are protective: If the oxide is porous enough for oxygen to get through, then no seal will be formed, and the metal beneath will continue to corrode. For example, magnesium oxide forms a layer with high surface porosity that does not halt corrosion. Oxygen molecules still flow through and react with the magnesium underneath.
Likewise, the elements in the surrounding environment also matter. Stainless steel, for example, can be challenged by salt or iron deposits. If the overall chemistry of the surface no longer passivates naturally, rust will set in.
Forced passivation
For some alloys, natural passivation can take a long time; for others, it can develop unevenly, with variations in the grain of the metal or in the presence of surface deposits. Metallurgists created active passivation methods to speed up and standardize the process to create immediately usable products.
Gun “bluing” was an early example of forced passivation through chemical manufacturing means. One of the oxides of iron is magnetite, a black oxide (Fe3O4), and this oxide does not flake the way rust (Fe3O3) will. Several chemical processes can be used to create these black oxides using heat and caustic solutions. However, although bluing does protect against corrosion, a damaged layer does not “heal” in normal conditions. Bluing is therefore a manufactured sealant that needs upkeep and care.
Today, common active passivation treatments have several steps:
- Cleaning the item to remove surface oil and impurities. There should be no areas sealed from the acid bath by an external coating.
- Passivating in baths of nitric acid or citric acid, or by using an electrochemical process. For stainless steel, this step removes any free deposits of iron that would inhibit the stainless steel from forming a solid passive film. On a microscopic level, a deposit of free iron would prevent the passive chromium oxide layer from forming a continuous seal. The passivation layer after the nitric acid bath is made of Cr2O3.
- Rinsing the item of all traces of the acid solution, taking any remaining free iron with it.
- The item is placed under conditions that promote oxidation. Conditions include a combination of increased temperature and humidity, and use of rust-promoting agents such as salt spray, copper sulphate, or potassium ferricyanide.

Stainless steel and other self-passivating alloys
Stainless steel is a tough metal that has corrosion resistance through natural passivation. Since its invention in 1913, many industries have come to rely on the metal. However, it is not guaranteed to be rust free.
Stainless steel, like other steels, is mainly made of iron and carbon. The revolutionary addition to the alloy is chromium. Chromium, exposed to air, quickly forms a passive oxide layer that seals the iron and protects it. Various grades of stainless steel contain different supporting metals: molybdenum, silicon, and other constituents all lend passive support for different applications. Some grades are superior at managing heat, others at resisting corrosion due to salt: the chemical mix of the alloy changes how it behaves under different conditions. Iron deposits, heat, contact with other metals, salt, and acids can all challenge the oxide layer.
Aluminum is another metal that naturally passivates. Aluminum oxide forms on most (although not all) aluminum alloys when exposed to air, making the surface self-protective. Aluminum oxide can be challenged by salt, electrochemical stress, or trapped moisture. With both stainless steel and aluminum, manufacturing processes are used to help support the creation of passive layers of a greater thickness or uniformity than might occur naturally.

Passivation standards for stainless steel and aluminum
Stainless steel and aluminum are both self-passivating materials, but they are not impervious to corrosion. Irregularities in grain, created by manufacture or heat treatments, can create weakness. Surface deposits of oil or other chemicals also may interrupt the passive film. To ensure the quality of stainless steel and aluminum grades, there are now standard passivation processes and tests.
ASTM specifications A380 and A967 set standards and quality-testing procedures for stainless steel passivation using nitric acid, citric acid, or by electrical means.
Chromium is sometimes used to passivate other materials, but often via application rather than being built into the alloy. A process called chromium conversion is used for aluminum and other metals, such as zinc and nickel. In this technique, a chromium gel is painted onto the metal surface. The chemical binds to the surface of the metal, creating a passive layer that demonstrates a high corrosion resistance. A scratch to a chromium conversion passivation will undergo a self-healing process. Chromium around the scratch moves in to bond and re-create the passivation layer. However, the scratch needs to be small enough in size that this is possible with surrounding chromium.

Pickling vs. passivation of stainless steel
The passivation of steel is a process that uses an acid bath to leave behind an oxide layer. Pickling is another acid-bath treatment, but it has the opposite goal: pickling uses an acid to clean oxides from the surface of the metal.
When oxides coat the surface of a metal, the item becomes more challenging to machine. Oxides cause greater stress on tool bits and can defeat attempts to seal the surface with paint or powder coating. Pickling removes all oxides, including any that work as a passive layer. Steel and iron are usually pickled.
In the production of a metal part, the part might be pickled, machined, and then passivated.
Electropolishing stainless steel and other metals
Electropolishing is a metal finishing step that deburrs and smooths, leaving a gleaming clean surface. It can be used on many metals, including those that are not encouraged to passivate, like copper. On passivating metal, a smooth surface can create unbroken, resilient passive layers.
An object to be electropolished is given a positive charge and dipped into an electrolyte bath. Surrounding cathodes pull the surface molecules from the object, shaving its top layer. Jagged protrusions are the first to be pulled away. On both macroscopic and microscopic levels, electropolished metal has few irregularities or crevices.
When electropolishing stainless steel, iron is preferentially removed, leaving more chromium at the surface. A smooth, uninterrupted surface free of iron naturally encourages a solid passivation layer on stainless steel.
When passivation fails
Passivation isn’t always the ideal solution; potential issues span a wide range of variables. Certain types of metals are unable to passivate, because the metal flakes as it corrodes. If a metal is structurally able to passivate, things can still go wrong during the acid bath process. On the other hand, even a perfectly passivated metal may not be usable in certain industries—the chemistry can cause issues in electrochemical applications.

Why does some metal flake as it corrodes?
Metal oxides can have a larger crystal structure than their constituent metal molecules. For example, iron oxide (III), the red corrosion byproduct better known as rust, has a larger structure, and therefore larger surface area, than the elemental iron that produces it. This larger surface area forces the oxide to lift up from the surface of the metal below, causing bubbling and flaking. Separation of the oxide from the metal exposes the next layer to air and moisture, and the cycle continues, eating the surface away.
In situations where oxides, hydroxides, or sulfides have a larger surface area than the metal they arise from, no passivation layer forms.
Passivation Flash Attacks
Sometimes, a manufacturer will find that in a batch of passivating items, one or more turn black and start etching even in a strong nitric acid bath. This active state is known as “flash attack.” It can be quite confusing, since sometimes it can attack some items but leave others in the same dip-basket alone.
Reasons for flash attack have to do with the consistency of the chemicals involved in making the passive film. If the nitric acid bath has been in use for a long time, it may have accumulated salts or water. The parts themselves are often a problem: there might be cutting oil left on machine parts, or heat treatments or heat control during machining may have unevenly changed the molecular structure of the part. There might also be inclusions or inconsistencies within the alloy itself.
Avoiding passivation
There are times where passivation can cause problems for the proper functioning of a metal part.
Electrochemical treatments often require metal cathodes and anodes to run an electrical current through a solution. These systems may encourage the formation of oxides that cling to the outside of the cathodes. As the cathodes become fouled with the oxides, the system becomes less effective.
In these systems, passivation is a problem. Switching polarity sometimes resolves the problem. Opposite pulses of electricity allow the oxides to fall away from the cathodes. The oxide sludge or slag can fall away and not interfere with the electrochemical activity of the process.

Passivation layers to seal and protect
Many metals react with the environment producing oxides, hydroxides, or sulfides. These products of corrosion arise in similar ways but have different properties.
Silver tarnish, arising slowly as silver combines with sulfides in the air, works as a passive layer. It dulls the surface of the metal, and is often polished away. In comparison, copper’s green patina, or verdigris, is often an aesthetic prize for the depth and range of green color it produces. Verdigris is a mixture of carbonates, sulfides, sulfates, and chlorides, created by copper reacting to acid rain or carbon dioxides. Rust, the most common iron oxide, produces orange or brick-red pigment. Unlike verdigris, it must be carefully controlled so that the bubbling surface does not allow the steel beneath to rust away.
Passivation layers seal and protect a metal object from further oxidization. With chromium-based passive layers, such as on stainless steel, this film is often thin enough that it does not change the surface look or function of the metal. Thin passivation often only changes the metal in very specific settings—such as welding, machining, or in electrochemical systems. The greatest benefit provided by self-passivating metals is the ability to “self-heal.” For maximum corrosion-protection, combine a self-passivating metal with an applied sealant like an oil, powder coating, or paint.












