What floats our boats?

Most metals on the market are not sold as objects made of only one element. To get material properties, metals are mixed with other elements to create alloys; specifications of these alloys are known as their grades. A manufacturer chooses a grade based on the final use of the product.
What is marine grade aluminum?
There are many grades of commercially available aluminum. Marine-grade aluminum are alloys that can handle exposure to sun and water due to the elements that are alloyed with the aluminum. Magnesium and silicon are the primary additions to bring the alloys up to marine grade. Some marine grades are more tolerant of salt water, which brings extra chemical stress to the metal.
Marine grade aluminum is used for boats, but can also be found on docks, railings, ladders, stairs, and other furnishings or items that are usually used on or near the water. Marine grades of aluminum can also be used in tanks or storage facilities: they are not exclusively used in marine situations.
Although aluminum is commonly used for floating watercraft, it is rarely used for submarines. The compression forces involved in diving wreak havoc on aluminum, causing eventual fatigue and cracking. Additionally, the constant exposure to sea water without air speeds corrosion.
What kind of aluminum is used in boats?
Picking the right grade of aluminum means balancing cost, ease of manufacturing, and the alloy’s material properties. Some alloys are easier to form or weld, which are important manufacturing considerations in hull manufacturing. Other alloys may provide superior corrosion resistance.
Aluminum boats for the retail market, including those by major manufacturers Lund and Princecraft, are primarily crafted from 5052. This wrought aluminum grade contains 2.2%-2.8% magnesium and 0.15%-0.35% chromium: these help it with corrosion resistance. The workability is only fair, however, and so some components on the boat may be made with other alloys.
6061, a builder’s aluminum alloy with additional silicon, carries excellent corrosion resistance and is easier to work, weld, and finish than the 5052. This aluminum is often used for extra strength or to form components that would be impossible to make with 5052. 6061 is more expensive and therefore is only used where the material properties and workability are worth the cost.
85% of aluminum is sold as wrought aluminum but cast aluminum may sometimes be used for specific components that need to be made in near-net shapes out of the mold. The most-used cast marine grade is A356, which is roughly equivalent to 6061. The silicon in the 6061 helps cast aluminum catch details in the mold.
Galvanic corrosion in aluminum
Marine vessels are often ornamented with brass, bronze, or copper. Stainless steel is often also used in marine environments, for its famed resistance to corrosion.
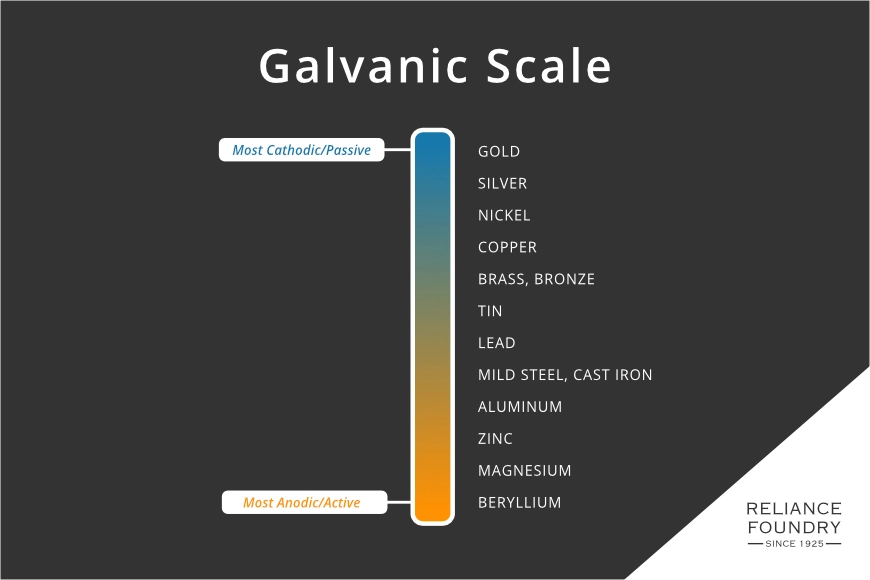
These metals should be avoided in any attachments to aluminum boats. They can cause galvanic corrosion. Galvanic corrosion happens when two metals, far apart on the galvanic scale, are in contact in a bath of electrolytes. The anodic or active element gives ions to the passive or cathodic metal, slowly eating away at the anode. Aluminum is one of the more active metals and will give up many ions to anything copper, and at a slower but still consistent rate to stainless.
Aluminum is so active that sometimes it is used as what’s known as a “sacrificial anode.” A piece of aluminum will be attached to the hull of a steel ship or dropped over the side in a net. The aluminum corrodes and helps protect the steel.
Galvanic corrosion is another reason submarines are not made of aluminum. Many of the components necessary to run a submarine—like a nuclear reactor—must be made with other metals that, while in contact, could cause quicker corrosion in the aluminum hull.
Painting (using epoxy paint) below the waterline is one way to lower the risk of galvanic corrosion, especially when moored close to steel hulled boats or submerged steel components.

Why use aluminum near water?
Aluminum may not be best for constant submersion, but it is popular for boats, docks, pontoons, site furniture and ladders because it is both light and strong. When raw aluminum is exposed to air and water (rather than constantly submerged) the aluminum oxide that develops on the surface can provide protection against further corrosion. The grade is important though: common 3000-series grades of aluminum can oxidize quickly in a marine environment. If you’re sourcing an aluminum item to go in or around water, make sure you’re getting something considered marine grade, either 5000 series or some of the 6000 series builder’s aluminum. With care and maintenance, these grades give long use life for aluminum objects near the water.












